|
Thus, the two electrons in the carbon 2 p orbitals have identical n, l, and m s quantum numbers and differ in their m l quantum number (in accord with the Pauli exclusion principle). The orbitals are filled as described by Hund’s rule: the lowest-energy configuration for an atom with electrons within a set of degenerate orbitals is that having the maximum number of unpaired electrons. We now have a choice of filling one of the 2 p orbitals and pairing the electrons or of leaving the electrons unpaired in two different, but degenerate, p orbitals. The remaining two electrons occupy the 2 p subshell. 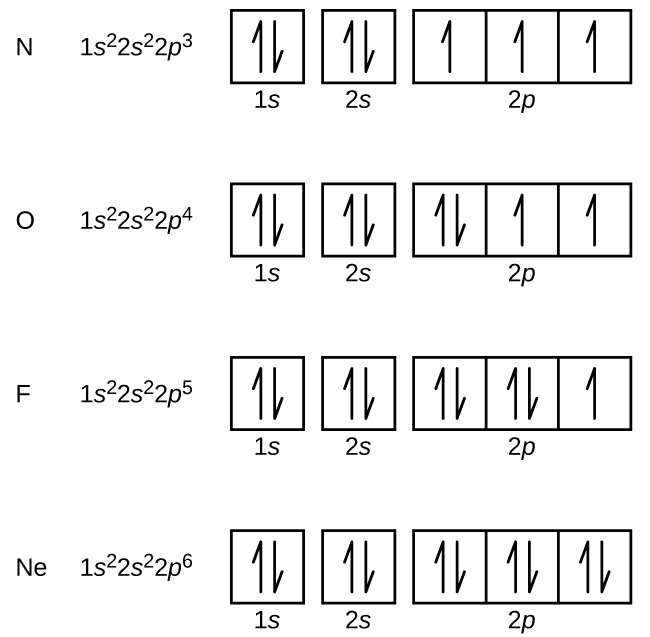
Using arrows to show the spin orientation of each electron, the orbital diagram is often shown this way: The single electrons in the two p-orbitals is in accordance with Hunds Rule. The total number of electrons in carbon is six. In the explanation below, I show a common means of diagramming this. Four of them fill the 1 s and 2 s orbitals. Carbon is the 6th element in the periodic table and its symbol is ‘C’. When drawing orbital diagrams, we include empty boxes to depict any empty orbitals in the same subshell that we are filling.Ĭarbon (atomic number 6) has six electrons. There are three degenerate 2 p orbitals ( m l = −1, 0, +1) and the electron can occupy any one of these p orbitals. 
Because any s subshell can contain only two electrons, the fifth electron must occupy the next energy level, which will be a 2 p orbital. 
The n = 1 shell is filled with two electrons and three electrons will occupy the n = 2 shell. The fourth electron fills the remaining space in the 2 s orbital.Īn atom of boron (atomic number 5) contains five electrons. Thus, the electron configuration and orbital diagram of lithium are:Īn atom of the alkaline earth metal beryllium, with an atomic number of 4, contains four protons in the nucleus and four electrons surrounding the nucleus. The first two electrons in lithium fill the 1 s orbital and have the same sets of four quantum numbers as the two electrons in helium.\) ). What is the orbital diagram for Carbon HUNDS RULE states that all orbitals in each sublevel must be filled, before the electrons will double up. The next atom is the alkali metal lithium with an atomic number of 3. The n = 1 shell is completely filled in a helium atom. When we get to N (Z 7), we have to put one electron into each of. The electron configuration and orbital diagram of helium are: The electrons in the 2p orbitals on carbon can therefore be represented as follows. For orbital diagrams, this means two arrows go in each box (representing two electrons in each orbital) and the arrows must point in opposite directions (representing paired spins). 
Write the electron configuration for each of the following ions: As 3+. To draw the orbital diagram, begin with the following observations: the first two electrons will pair up in the 1s orbital the next two electrons will pair up in the 2s orbital. Draw the orbital energy diagram, for Na and S-2, putting the valence. The electron configuration can be written as 1s 2 2s 2 2p 4. Orbital diagrams: pictorial representations of the electron configuration. Consider also the electron configuration of oxygen. Atomic valence electrons (shown in boxes on the left and right) fill the lower-energy molecular orbitals before the higher ones, just as is the case for atomic. Suppose we want to draw the orbital-box diagram of carbon: Step 1. The electron configuration for carbon is 1s22s22p2. Carbon (atomic number 6) has six electrons. This scheme of bonding and antibonding orbitals is usually depicted by a molecular orbital diagram such as the one shown here for the dihydrogen ion H 2 +. Electron configurations tells us in which orbitals the electrons for an element are located. This diagram shows how the electrons in the carbon atom are arranged in different orbitals. This is in accord with the Pauli exclusion principle: No two electrons in the same atom can have the same set of four quantum numbers. Write the number of valence electrons, electron configuration, and orbital diagram of the following element: Phosphorus i) number of valence electrons: ii) total electrons on level n 3: iii) electron configuration: iv) orbital diagram: Give the noble gas electron configuration of carbon (C). The orbital diagram, the electron configuration and the energy diagram. The carbon orbital diagram is a graphical representation of the electron configuration of the carbon atom.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
